 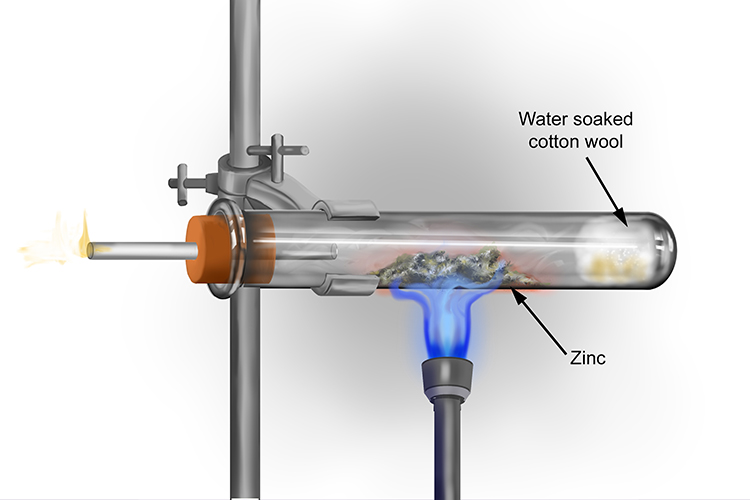 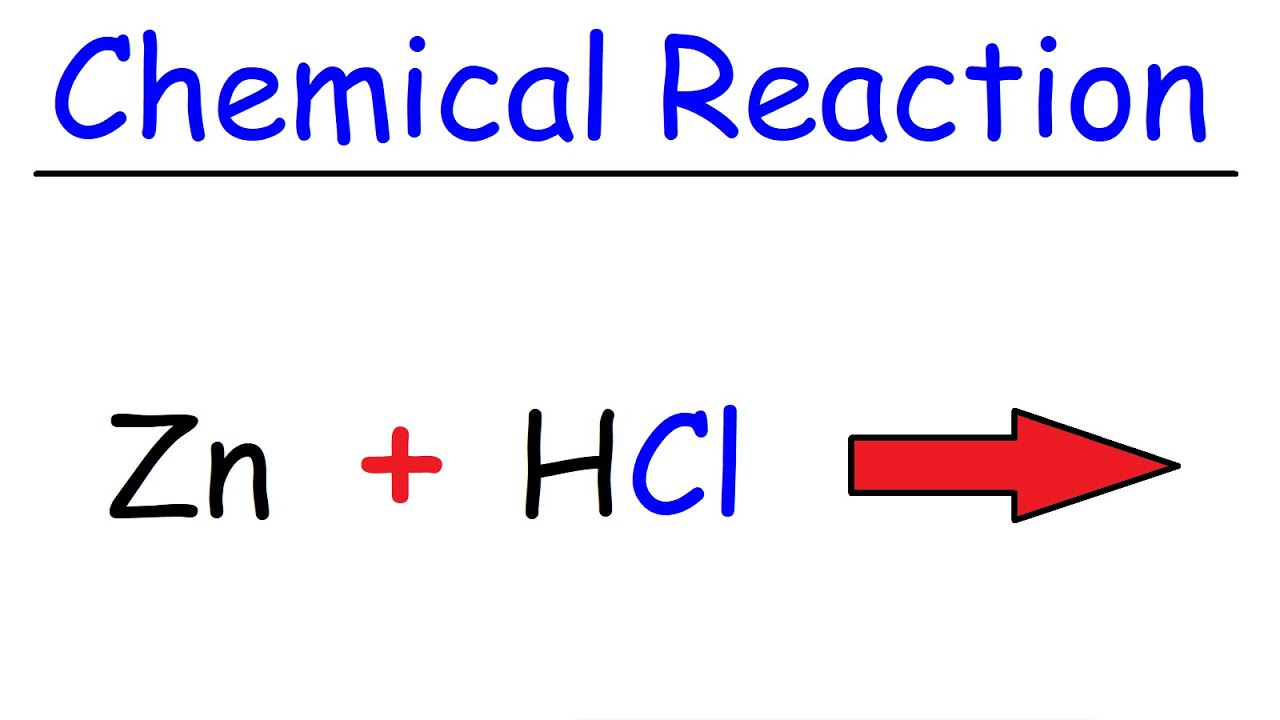
This work reveals important factors that govern the reactivity of zinc-bound cysteines in these two structural classes of zinc fingers and can be used to identify reactive zinc fingers in proteins. and to promote stain-blocking (zinc oxide’s reactivity produces cationic ions which chemically interact with anionic stains.) Unfortunately, as an additive, it does not. Indeed the entropies of activation are strongly linked to the conformational behavior of the peptide in solution. Metal X lies between zinc and iron in the reactivity series.Which statements about metal X are correcta.It reacts with steam to produce hydrogen gas.b. As described earlier elsewhere, Zinc Oxide is very reactive and will quickly form metallic soaps from free fatty acids found in oil paints. Interestingly, we observe that the more flexible zinc finger sites are the more reactive. Neutral Zn(Cys) 2(His) 2 cores are intrinsically less reactive than negatively charged Zn(Cys) 4 and Zn(Cys) 3(His). During the initial stages of the fuel cycle, the concentration is relatively high (10002500 ppm boron) and then is gradually reduced to 0 ppm. In all cases, the reaction is characterized by a small enthalpy of activation, which shows that the nucleophilic reaction of zinc-bound cysteines is easy, and large unfavorable negative entropy of activation. The rate constants of the reaction of the nine zinc finger models with H 2O 2 were measured at various temperatures to determine the activation parameters. The coordination properties and the structural behaviors of the new members of the latter family were carefully characterized. One family is derived from the consensus peptide of classical ββα zinc fingers and the other one derived from the zinc finger site of the oxidative stress sensor Hsp33 that adopts a loosened zinc ribbon fold. We use a set of nine peptides belonging to two different peptide families with (Cys) 4, (Cys) 3(His) and (Cys) 2(His) 2 coordination sets. In this article, we investigate the nucleophilic reaction of zinc finger model peptides with H 2O 2 in order to get deeper insight into the factors governing the reactivity of zinc-bound cysteines of zinc finger sites. Na + ROH RONa + 1/2 H 2, in which R is the organic portion of the alcohol (R CH 3 for methanol, CH 3 CH 2 for ethanol, etc.). Sodium reacts with anhydrous alcohols to form the respective alcoholates (or alkoxides) according to. However, the nucleophilic reactivity of zinc fingers remains poorly understood. The organic reactions of sodium have been studied to a greater extent than those of any of the other alkali metals. Electrophiles targeting especially vulnerable structural zinc fingers have also been proposed as therapeutic agents against cancers or HIV. The two H free radicals combine and form H2 gas. The new unstable compound ZnCl reacts almost instantly with another HCl, forming ZnCl2 and releasing another H free radical. This happens when the Zn and HCl collide and release a H free radical. During the past decades it has been established that zinc-bound cysteines in proteins can react with various electrophiles to perform physiological functions such as alkyl transfer reactions or oxidative stress sensing. Actually what happens is that the Zinc displaces the H-atoms from 2 HCl molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
